ANSI/ADA 128-2022: Hydrocolloid Impression Materials
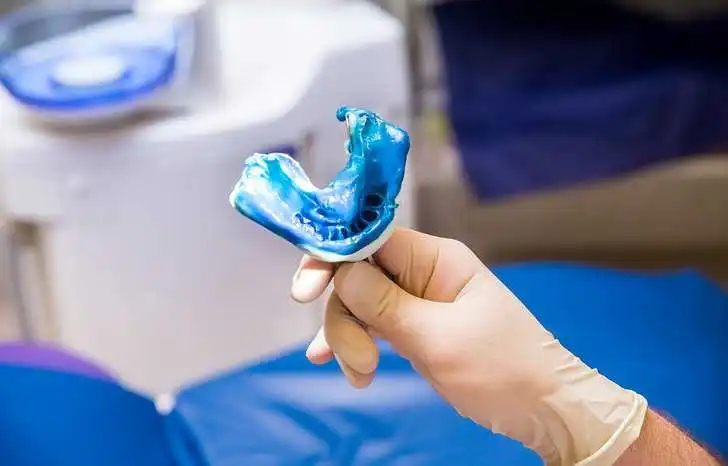
Hydrocolloids were the first elastic impression materials available to dentists. When mixed, they form a viscous liquid that can be seated over oral structures. The liquid sets, becoming a gelatin-like solid that is flexible enough to be drawn over undercuts without significant permanent deformation. Hydrocolloids are important materials used in dentistry due to their biocompatibility with tissues, ease of use, physical properties, and hydrophilicity with oral tissues. ANSI/ADA 128-2022: Dentistry-Hydrocolloid Impression Materials specifies the requirements and test methods for hydrocolloid impression materials.
Dental Impression Materials
Impression materials are classified as either non-elastic or elastic based on mechanical properties. Non-elastic or rigid impression materials include impression compounds, zinc oxide eugenol, and impression waxes. Meanwhile, elastic materials are capable of stretching, compressing, and recovering after deformation; they comprise reversible and irreversible hydrocolloids, addition and condensation silicones, polysulfides, and polyether.
In dentistry, most intricate and precise procedures are made of hydrocolloids due to the material’s characteristics. Hydrocolloids are complex polysaccharides that disperse or dissolve in aqueous solution to give thickened or vicious effects, and they possess high molecular weight. They are found in the simplest to most complex material such as impression making, fillings, separating media, electro-polishing etc.
What Is ANSI/ADA 128?
ANSI/ADA 128-2022 specifies requirements and test methods for amalgam separators used in connection with dental equipment in the dental treatment center. The American National Standard specifies the efficiency of the amalgam separators in terms of the level of retention of amalgam and the test procedure for determining this efficiency. It also helps to determine whether elastic aqueous agar and alginate hydrocolloid dental impression materials are of the quality needed for their intended purposes. ANSI/ADA 128-2022 details the requirements for labeling and instructions for use.
This standard does not address possible biological hazards associated with the materials. Assessment of these hazards is addressed in ANSI/ADA 41, ISO 7405 and the ISO 10993 series.
Agar Hydrocolloid Impression Materials
The two common hydrocolloids widely used in dentistry are reversible (agar) and irreversible (alginate) materials. Reversible hydrocolloid impression materials are formulated with agar and solidify from their liquid state to a gel in the mouth to capture a highly accurate impression. These materials are temperature sensitive, changing from liquid to solid at different temperatures. As a result, they are often used with special water-cooled trays in order to capture the most accurate impressions. Because of the reversible nature of these materials, models should be poured immediately after the impression is taken. It is usually only possible to pour one model per impression, which means reversible hydrocolloids need to be used under proper conditions and models must be created immediately after the impression is captured.
Classification of Agar Hydrocolloid Impression Materials
The agar impression materials in ANSI/ADA 128-2022 are classified according to the consistencies they exhibit when they are ready for impressing against the oral or craniofacial tissue surfaces, and when tested.
- Type: heavy bodied, for making impressions of complete or partial dental arches, with or without the use of companion increments of lighter bodied Type 2 or Type 3 agar impression materials.
- Type 2: medium bodied, for making impressions of complete and partial dental arches, with or without the use of companion syringe-extruded increments of Type 3 agar materials.
- Type 3: light bodied, for syringe use with either the Type 1 or Type 2 agar materials.
- Type 3A: light bodied, material formulated for syringe use in a reversible/non-reversible impression material system, and that has been claimed to be capable of bonding to a companion alginate impression material that will make up the greater part of an agar/alginate impression material system.
ANSI/ADA 128-2022: Dentistry-Hydrocolloid Impression Materials is available on the ANSI Webstore.