The Royal Swedish Academy of Sciences awarded the 2019 Nobel Prize in Chemistry to three scientists who developed lithium-ion batteries: John B. Goodenough, M. Stanley Whittingham, and Akira Yoshino.
Why the Nobel Prize for Lithium-Ion Batteries is So Important
Any Nobel Prize-winning scientific effort is monumental, but this specific award stands out due to the near-ubiquity of the work. If you’re reading this on a cell phone or laptop computer, you are using a lithium-ion battery. Beyond these everyday items, these batteries power electric cars, aid medical devices, and even help store the energy absorbed by solar photovoltaic panels. As noted by the Royal Swedish Academy of Sciences:
“Lithium-ion batteries have revolutionised our lives since they first entered the market in 1991. They have laid the foundation of a wireless, fossil fuel-free society, and are of the greatest benefit to humankind.”
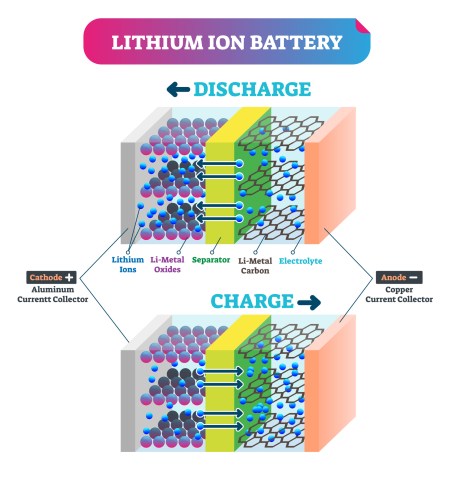
Lithium-ion batteries aren’t the backbone of modern society; they’re the heart. As simple as generating power through the flow of ions from anode to cathode (and vice versa when charging), lithium ion batteries, which can be recharged hundreds of times, had to undergo notable developments by these three scientists before they could become commonplace.
The Development of the Lithium-Ion Battery
While awarded the Nobel in 2019, lithium-ion batteries are by no means a new invention. While the battery itself traces its roots to 1800, when Allessandro Volta stacked discs of copper and zinc and linked them with a cloth soaked in salty water, the lithium ion battery’s development commenced in the 1970s.
During the oil crisis, numerous scientists worked to reduce society’s dependence on fossil fuels. Wittingham discovered that titanium disulfide had a molecular structure that permitted lithium-ions into small pockets. His work resulted in the first functional lithium ion battery, which stored 10 times as much energy as the average lead-acid battery while being lightweight and requiring little maintenance. Unfortunately, when repeatedly charged, thin strands of metallic lithium would extend out from the negative electrode, occasionally reaching the cathode, short circuiting the battery and potentially leading to an explosion.
Goodenough, noticing that cobalt oxide was similar in structure to titanium disulfide, improved upon the battery. His material could tolerate lithium being pushed into it and pulled continuously. In result, his lithium ion battery, generating four volts, was twice as powerful as the one constructed by Wittingham.
Yoshino later demonstrated that more complicated carbon-based electrodes could house lithium-ions in between their layers. This eliminated pure lithium from the battery, using only lithium-ions.
Lithium-ion was commercialized in 1991 by Sony. Over the past thirty years, due to its compact nature and reliability, lithium-ion batteries have remained crucial to consumer electronics. They see numerous mentions among standards throughout industries and are the focus of NEMA standard ANSI C18.2M. It’s a challenge to imagine a world without them.
Congratulations to the winners! To celebrate, devote some time to just you and your smartphone (but you’re probably doing that already).
You can learn more about the other Nobel Prizes and Laureates here, which are awarded for “the greatest benefit to mankind.”